Which Best Explains Why Changes To The Atomic Theory Were Necessary?
2.1: The Diminutive Theory of Matter
- Folio ID
- 21697
Learning Objectives
- Explain how all matter is composed of atoms.
- Describe the modern atomic theory.
Take some aluminum foil. Cut it in half. Now there are two smaller pieces of aluminum foil. Cutting one of the pieces in half again. Cutting ane of those smaller pieces in half again. Go along cutting, making smaller and smaller pieces of aluminum foil. It should be obvious that the pieces are still aluminum foil; they are just condign smaller and smaller. But how far tin can this exercise exist taken, at least in theory? Can one continue cutting the aluminum foil into halves forever, making smaller and smaller pieces? Or is there some limit, some absolute smallest piece of aluminum foil? Thought experiments like this—and the conclusions based on them—were debated as far dorsum as the fifth century BC.
John Dalton (1766-1844) is the scientist credited for proposing the diminutive theory. The theory explains several concepts that are relevant in the appreciable world: the composition of a pure gilt necklace, what makes the pure gold necklace different than a pure silver necklace, and what occurs when pure gold is mixed with pure copper. This section explains the theories that Dalton used as a basis for his theory: (1) Law of Conservation of Mass (two) Police of Definite Proportions, and (3) Constabulary of Multiple Proportions
Police force one: The Conservation of Mass
"Nix comes from goose egg" is an important idea in ancient Greek philosophy that argues that what exists at present has always existed, since no new matter can come into beingness where there was none before. Antoine Lavoisier (1743-1794) restated this principle for chemistry with the police force of conservation of mass, which "means that the atoms of an object cannot be created or destroyed, but tin be moved around and be changed into different particles." This police says that when a chemical reaction rearranges atoms into a new product, the mass of the reactants (chemicals earlier the chemical reaction) is the same as the mass of the products (the new chemicals fabricated). More simply, whatever you practise, y'all volition however have the same amount of stuff (still, certain nuclear reactions like fusion and fission tin convert a small function of the mass into free energy.
The constabulary of conservation of mass states that the full mass present before a chemical reaction is the same as the full mass nowadays afterwards the chemical reaction; in other words, mass is conserved. The law of conservation of mass was formulated by Lavoisier equally a outcome of his combustion experiment, in which he observed that the mass of his original substance—a drinking glass vessel, tin, and air—was equal to the mass of the produced substance—the glass vessel, "tin calx", and the remaining air.
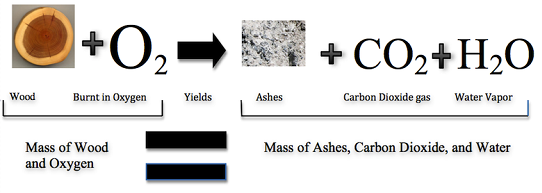
Historically, this was a difficult concept for scientists to grasp. If this law was true, then how could a large piece of woods be reduced to a pocket-size pile of ashes? The wood clearly has a greater mass than the ashes. From this ascertainment scientists ended that mass had been lost. Notwithstanding, Figure \(\PageIndex{ane}\) shows that the called-for of word does follow the law of conservation of mass. Scientists did non account for the gases that play a disquisitional role in this reaction.
The police force of conservation of mass states that the total mass present earlier a chemical reaction is the same as the total mass present after the chemical reaction.
Law ii: Definite Proportions
Joseph Proust (1754-1826) formulated the police force of definite proportions (also chosen the Law of Constant Composition or Proust'due south Police force). This law states that if a compound is broken downwards into its elective elements, the masses of the constituents will always have the same proportions, regardless of the quantity or source of the original substance. Joseph Proust based this law primarily on his experiments with basic copper carbonate. The illustration below depicts this constabulary in action.
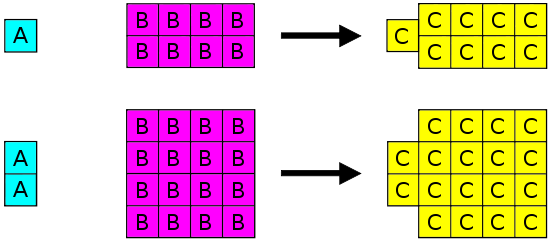
Constabulary of Definite Proportions states that in a given blazon of chemical substance, the elements are always combined in the same proportions by mass.
The Police force of Definite Proportions applies when elements are reacted together to form the same product. Therefore, while the Law of Definite Proportions can be used to compare two experiments in which hydrogen and oxygen react to form water, the Law of Definite Proportions can not be used to compare one experiment in which hydrogen and oxygen react to course water, and another experiment in which hydrogen and oxygen react to form hydrogen peroxide (peroxide is some other material that tin be made from hydrogen and oxygen).
Case \(\PageIndex{ane}\): water
Oxygen makes upward 88.8% of the mass of any sample of pure water, while hydrogen makes up the remaining 11.2% of the mass. Yous can become h2o past melting water ice or snow, by condensing steam, from river, ocean, pond, etc. It tin exist from different places: USA, Great britain, Australia, or anywhere. It can be made by chemical reactions similar burning hydrogen in oxygen.
However, if the h2o is pure, it will always consist of 88.eight % oxygen by mass and 11.2 % hydrogen by mass, irrespective of its source or method of grooming.
Law iii: Multiple Proportions
Many combinations of elements tin can react to form more than one chemical compound. In such cases, this police states that the weights of one element that combine with a stock-still weight of another of these elements are integer multiples of one another. It'due south easy to say this, just please make sure that y'all understand how it works. Nitrogen forms a very large number of oxides, five of which are shown here.
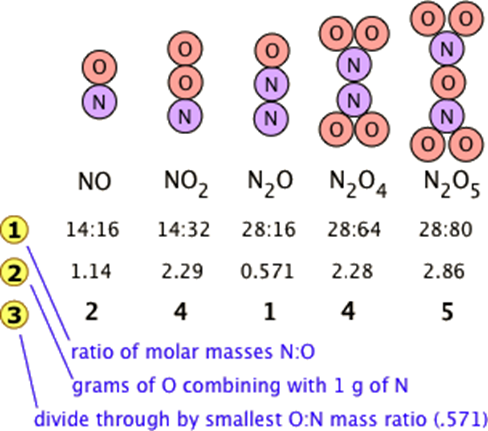
The law of multiple proportions states that if two elements form more than one chemical compound between them, the masses of i element combined with a stock-still mass of the second element grade in ratios of small integers.
Example \(\PageIndex{2}\): Oxides of Carbon
Consider 2 divide compounds are formed by simply carbon and oxygen. The offset compound contains 42.nine% carbon and 57.one% oxygen (by mass) and the 2nd chemical compound contains 27.three% carbon and 72.7% oxygen (again past mass). Is this consistent with the law of multiple proportions?
Solution
The Law of Multiple Proportions states that the masses of 1 chemical element which combine with a stock-still mass of the second element are in a ratio of whole numbers. Hence, the masses of oxygen in the two compounds that combine with a fixed mass of carbon should exist in a whole-number ratio.
Thus for every 1 g of the offset compound at that place are 0.57 g of oxygen and 0.429 g of carbon. The mass of oxygen per gram carbon is:
\[ \dfrac{0.571\; \text{yard oxygen}}{0.429 \;\text{g carbon}} = i.33\; \dfrac{\text{g oxygen}}{\text{g carbon}}\nonumber\]
Similarly, for i k of the 2d compound, there are 0.727 g oxygen and 0.273 g of carbon. The ration of mass of oxygen per gram of carbon is
\[ \dfrac{0.727\; \text{1000 oxygen}}{0.273 \;\text{yard carbon}} = 2.66\; \dfrac{\text{g oxygen}}{\text{thousand carbon}}\nonumber\]
Dividing the mass of oxygen per k of carbon of the second compound:
\[\dfrac{ii.66}{1.33} = 2\nonumber\]
Hence the masses of oxygen combine with carbon in a 2:1 ratio which s consistent with the Law of Multiple Proportions since they are whole numbers.
Dalton'south Atomic Theory
The modern diminutive theory, proposed about 1803 past the English chemist John Dalton (Figure \(\PageIndex{iv}\)), is a key concept that states that all elements are composed of atoms. Previously, an cantlet was divers as the smallest part of an element that maintains the identity of that element. Individual atoms are extremely small; even the largest cantlet has an approximate diameter of only v.4 × 10−10 m. With that size, it takes over eighteen million of these atoms, lined up adjacent, to equal the width of the homo pinkie (most 1 cm).

Dalton's ideas are called the modernistic atomic theory because the concept of atoms is very old. The Greek philosophers Leucippus and Democritus originally introduced atomic concepts in the fifth century BC. (The give-and-take atom comes from the Greek word atomos, which means "indivisible" or "uncuttable.") Dalton had something that the ancient Greek philosophers didn't have, however; he had experimental evidence, such as the formulas of elementary chemicals and the beliefs of gases. In the 150 years or so before Dalton, natural philosophy had been maturing into modern science, and the scientific method was being used to study nature. When Dalton announced a modern atomic theory, he was proposing a fundamental theory to describe many previous observations of the natural earth; he was not only participating in a philosophical word.
Dalton's Theory was a powerful development as information technology explained the three laws of chemic combination (above) and recognized a workable distinction betwixt the fundamental particle of an chemical element (atom) and that of a compound (molecule). Six postulates are involved in Dalton'due south Atomic Theory:
- All matter consists of indivisible particles called atoms.
- Atoms of the same element are similar in shape and mass, but differ from the atoms of other elements.
- Atoms cannot be created or destroyed.
- Atoms of dissimilar elements may combine with each other in a fixed, unproblematic, whole number ratios to form compound atoms.
- Atoms of aforementioned element can combine in more than 1 ratio to form ii or more compounds.
- The atom is the smallest unit of matter that tin can take part in a chemical reaction.
In light of the electric current state of noesis in the field of Chemical science, Dalton'southward theory had a few drawbacks. According to Dalton'southward postulates,
- The indivisibility of an atom was proved wrong: an atom can be further subdivided into protons, neutrons and electrons. However an atom is the smallest particle that takes office in chemical reactions.
- According to Dalton, the atoms of aforementioned element are similar in all respects. Notwithstanding, atoms of some elements vary in their masses and densities. These atoms of different masses are chosen isotopes. For example, chlorine has ii isotopes with mass numbers 35 and 37.
- Dalton also claimed that atoms of different elements are different in all respects. This has been proven wrong in sure cases: argon and calcium atoms each have an same atomic mass (40 amu).
- According to Dalton, atoms of different elements combine in elementary whole number ratios to form compounds. This is not observed in complex organic compounds like saccharide (\(C_{12}H_{22}O_{11}\)).
- The theory fails to explicate the existence of allotropes (different forms of pure elements); it does not account for differences in backdrop of charcoal, graphite, diamond.
Despite these drawbacks, the importance of Dalton's theory should non be underestimated. He displayed exceptional insight into the nature of matter. and his ideas provided a framework that was afterwards modified and expanded by other. Consequentiually, John Dalton is often considered to be the father of modernistic atomic theory.
Fundamental Experiments in Chemistry: https://youtu.be/IhqqLGKmah4
References
- Petrucci, Ralph, William Harwood, Geoffrey Herring, and Jeffry Madura. General Chemistry. 9th ed. Upper Saddle River, New Jersey: Pearson Prentince Hall, 2007
- Moore, John. Chemistry for Dummies. John Wiley & Sons Inc, 2002.
- Asimov, Isaac. A Short History of Chemistry. , CT.: Greenwood Printing, 1965.
- Patterson, Elizabeth C. John Dalton and the Atomic Theory. Garden City, NY: Doubleday, 1970
- Myers, Richard. The Basics of Chemistry. Greenwood, 2003
- Demtröder, Wolfgang. Atoms, Molecules and Photons: An Introduction to Diminutive- Molecular- and Breakthrough Physics. 1st ed. Springer. 2002
Summary
This article explains the theories that Dalton used every bit a basis for his theory: (i) the Law of Conservation of Mass, (2) the Law of Constant Composition, (3) the Law of Multiple Proportions.
Source: https://chem.libretexts.org/Bookshelves/General_Chemistry/Map%3A_Chemistry_-_The_Central_Science_(Brown_et_al.)/02._Atoms_Molecules_and_Ions/2.1%3A_The_Atomic_Theory_of_Matter
Posted by: robinsonboweache.blogspot.com


0 Response to "Which Best Explains Why Changes To The Atomic Theory Were Necessary?"
Post a Comment